Bone Density Drug: Evidence and Safety Considerations
Medicines used to protect bone strength can reduce fracture risk for many people, but their benefits, limits, and safety profiles vary. Understanding the evidence behind these drugs, along with common side effects and rare complications, can help patients have clearer, more informed discussions with their clinician.
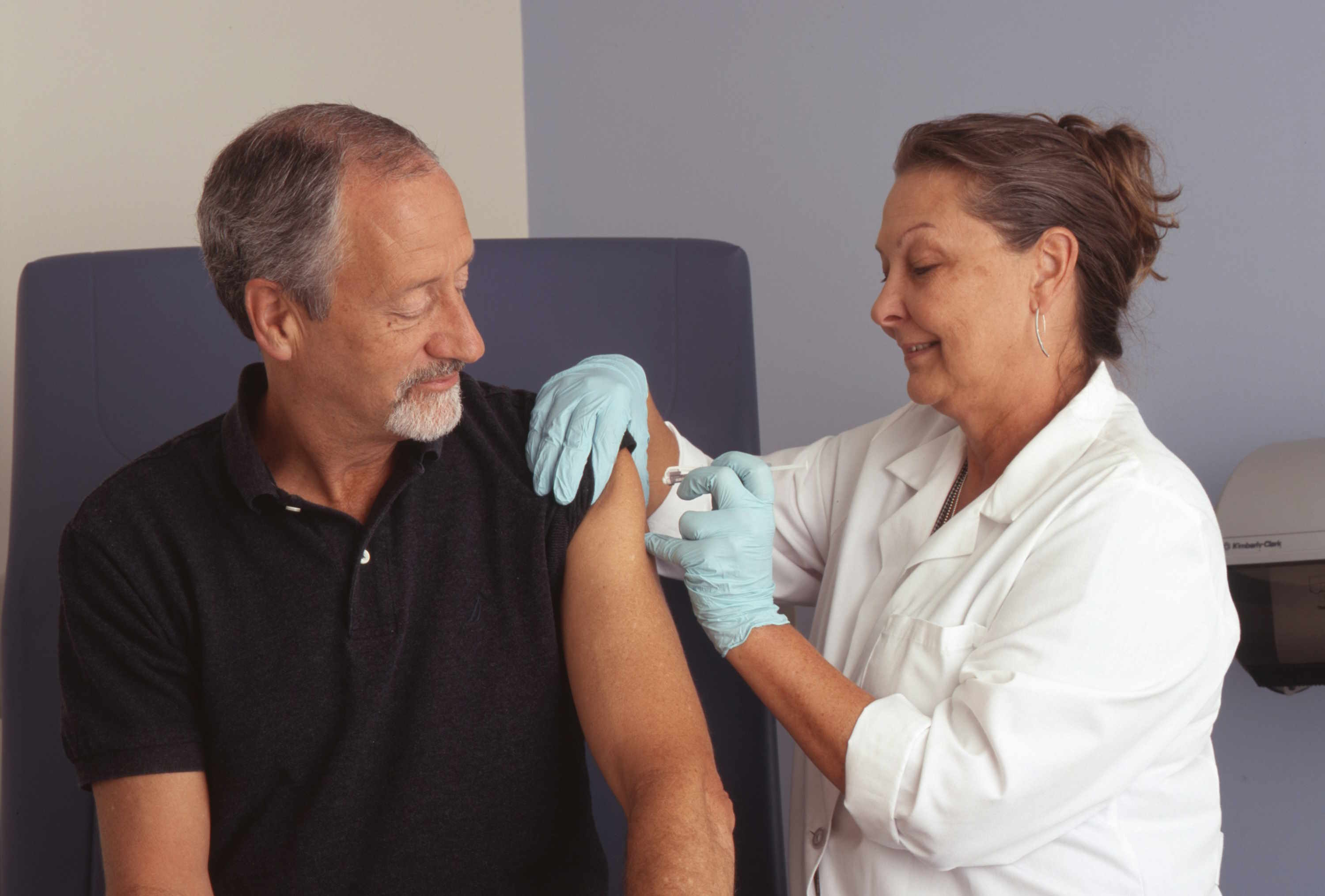
Medication for low bone strength is usually considered when fracture risk is high, imaging shows significant bone loss, or a person has already had a fragility fracture. These drugs do not work in exactly the same way, and they are not equally suitable for every patient. Age, kidney function, dental health, previous fractures, other illnesses, and the ability to take a medicine regularly all affect how treatment decisions are made. A balanced review of evidence and safety helps place potential benefits and harms in proper context.
This article is for informational purposes only and should not be considered medical advice. Please consult a qualified healthcare professional for personalized guidance and treatment.
What does the evidence show?
Clinical evidence shows that several bone medications can reduce the risk of fractures, especially in people with osteoporosis or a prior low-trauma fracture. The strongest evidence usually concerns vertebral and hip fracture prevention, although results differ by drug and patient group. Benefits are generally most meaningful in people at higher baseline risk, which is why treatment decisions often rely on formal fracture-risk assessment rather than bone scan results alone. Evidence is strongest when medicines are paired with adequate calcium, vitamin D, fall prevention, and follow-up monitoring.
Which drug classes are commonly used?
Common options include bisphosphonates, denosumab, selective estrogen receptor modulators, parathyroid hormone analogs, and sclerostin inhibitors. Some medicines mainly slow bone breakdown, while others stimulate new bone formation before switching to an antiresorptive approach. This distinction matters because treatment goals differ. A person with mild bone loss may need a different strategy than someone with multiple spinal fractures. Route of administration also varies, including tablets, injections every few months, or less frequent infusions, which can influence adherence and convenience.
Common and mild side effects
Common and mild side effects often depend on the specific drug. Oral bisphosphonates may cause gastrointestinal symptoms such as heartburn, throat irritation, nausea, or stomach discomfort, especially if dosing instructions are not followed carefully. Some patients also report muscle aches, joint pain, or headache. Injectable or infusion-based drugs can cause short-lived flu-like symptoms after treatment. These effects are often manageable, but they still matter because discomfort can make long-term treatment harder to continue. Mild side effects should be discussed if they persist or interfere with daily life.
Serious but rare adverse reactions
Serious but rare adverse reactions receive significant attention because they are concerning, even though they occur infrequently. Examples include osteonecrosis of the jaw and atypical femur fractures, both of which are uncommon but clinically important. Certain medicines may also be linked to low calcium levels, severe bone pain, or unusual fracture patterns after prolonged use. Risk tends to rise in specific situations, such as extended treatment duration, invasive dental procedures, very low calcium intake, or underlying medical conditions. Because these events are rare, their possibility must be weighed against the much more common risk of osteoporotic fractures.
Bone Density Drug: Risks, Side Effects, and Questions for Your Clinician
A useful clinical discussion often starts with a few practical questions: What is my actual fracture risk over the next several years? Which drug has the best evidence for my age and health profile? How long is treatment usually continued before reassessment? What side effects should prompt urgent review, and which are expected to settle? It is also reasonable to ask about kidney function, calcium and vitamin D status, planned dental work, and whether a pause or drug holiday is ever appropriate. These questions help personalize treatment rather than treating all patients as the same.
Bone Density Drug: Risks, Side Effects, and Clinical Considerations
Clinical considerations go beyond the medicine itself. Bone health management is strongest when medication is combined with weight-bearing activity, fall-risk reduction, smoking cessation, moderation of alcohol use, and attention to nutrition. Monitoring may include repeat bone density scans, review of fractures, blood tests, and reassessment of treatment response. In some cases, stopping therapy suddenly is not advisable, while in others long-term continuous treatment may not be ideal. The safest approach is individualized follow-up that accounts for changing risk over time rather than a one-time prescription decision.
No bone medication is completely risk free, but neither is untreated osteoporosis or severe bone loss. The key issue is proportionality: the people most likely to benefit are often those most at risk of disabling fracture. Evidence supports the use of several drug classes in appropriately selected patients, while safety considerations remind clinicians and patients to choose carefully, monitor consistently, and revisit the plan as circumstances change. Clear expectations and regular review are central to making treatment both effective and safer over the long term.